Research Themes
Chemotactic guidance signals for T cells at immune effector sites
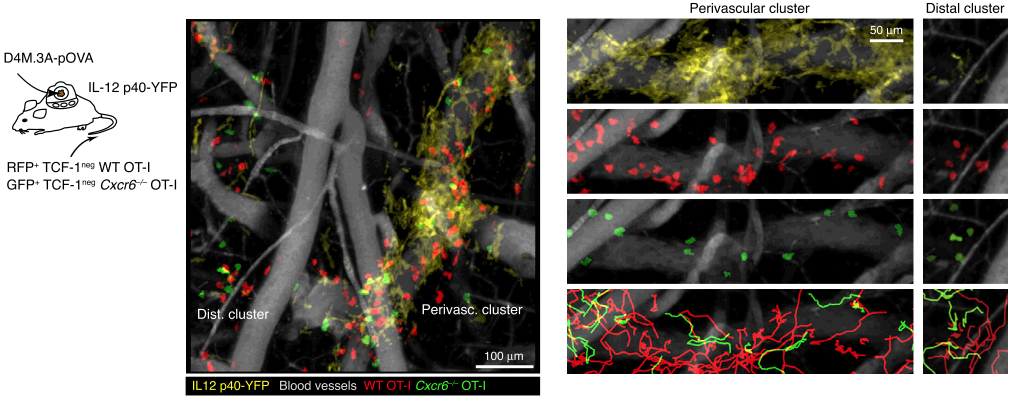 Antigenic priming of naive CD8+ T cells in secondary lymphoid organs (SLOs) produces short-lived effector as well as stemlike memory precursor cells. Both can egress from SLOs and via the bloodstream enter effector sites of immune responses, such as solid tumors. While tumor-infiltrating effector cells rapidly undergo terminal differentiation and lose effector activity during continuous antigen encounter, stemlike precursor cells retain self-renewal capacity but can also give rise to new effector cells, thereby locally replenishing the CD8+ T cell response at effector sites.
We have reported here that the conversion of stemlike into effector CD8+ T cells in the TME entails a major chemotactic reprogramming event that includes down-regulation of the chemokine receptor CXCR3 and upregulation of CXCR6. The latter positions effector cells near dense clusters of CCR7+, activated dendritic cells that express the CXCR6-ligand CXCL16, and which occupy perivascular immune niches in the tumor stroma that form under strongly inflammatory conditions favoring immunological control or rejection of tumor growth. CCR7+ dendritic cells trans-present IL-15 cytokine to effector T cells and thereby enable their rapid proliferative expansion in the TME, leading to more effective anti-tumor immunity. In collaboration with the Kobold lab we also reported here that ectopic expression of CXCR6 greatly amplified the therapeutic activity of CAR T cells against solid tumors in preclinical models.
In ongoing studies, we aim to understand the cellular and molecular mechanisms that organize perivascular immune niches of the tumor stroma, and how chemokines orchestrate T cell recruitment to, migration within, and egress from these niches.
Antigenic priming of naive CD8+ T cells in secondary lymphoid organs (SLOs) produces short-lived effector as well as stemlike memory precursor cells. Both can egress from SLOs and via the bloodstream enter effector sites of immune responses, such as solid tumors. While tumor-infiltrating effector cells rapidly undergo terminal differentiation and lose effector activity during continuous antigen encounter, stemlike precursor cells retain self-renewal capacity but can also give rise to new effector cells, thereby locally replenishing the CD8+ T cell response at effector sites.
We have reported here that the conversion of stemlike into effector CD8+ T cells in the TME entails a major chemotactic reprogramming event that includes down-regulation of the chemokine receptor CXCR3 and upregulation of CXCR6. The latter positions effector cells near dense clusters of CCR7+, activated dendritic cells that express the CXCR6-ligand CXCL16, and which occupy perivascular immune niches in the tumor stroma that form under strongly inflammatory conditions favoring immunological control or rejection of tumor growth. CCR7+ dendritic cells trans-present IL-15 cytokine to effector T cells and thereby enable their rapid proliferative expansion in the TME, leading to more effective anti-tumor immunity. In collaboration with the Kobold lab we also reported here that ectopic expression of CXCR6 greatly amplified the therapeutic activity of CAR T cells against solid tumors in preclinical models.
In ongoing studies, we aim to understand the cellular and molecular mechanisms that organize perivascular immune niches of the tumor stroma, and how chemokines orchestrate T cell recruitment to, migration within, and egress from these niches.
Conversion of immune-suppressive T regulatory into proinflammatory effector cells
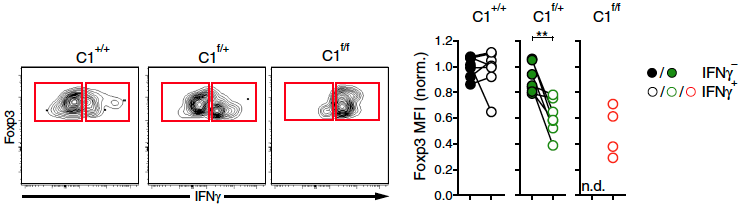 While T regulatory (Treg) cells are essential to maintaining immune homeostasis, they also limit spontaneous and therapeutically enhanced anti-tumor immune responses in cancer patients. Recent studies suggest that at sites of chronic inflammation, such as the tumor microenvironments (TMEs) of solid tumors, a small fraction of Treg cells can be induced to secrete the cytokine IFN-, and thereby contribute to the local tissue inflammation that renders some tumors susceptible to immune checkpoint blockade and other forms of cancer immunotherapy. Our prior studies have revealed that disruption of the Card11/Bcl10/Malt1 signalosome complex amplifies Treg conversion selectively in the TME, which you can read about here and here. Based on these findings, a small molecule inhibitor of Malt1 has now recently entered clinical testing as a cancer treatment.
We are now seeking to identify the local environmental factors, the signaling events, and the epigenetic changes that underlie this localized pro-inflammatory conversion of Treg cells.
While T regulatory (Treg) cells are essential to maintaining immune homeostasis, they also limit spontaneous and therapeutically enhanced anti-tumor immune responses in cancer patients. Recent studies suggest that at sites of chronic inflammation, such as the tumor microenvironments (TMEs) of solid tumors, a small fraction of Treg cells can be induced to secrete the cytokine IFN-, and thereby contribute to the local tissue inflammation that renders some tumors susceptible to immune checkpoint blockade and other forms of cancer immunotherapy. Our prior studies have revealed that disruption of the Card11/Bcl10/Malt1 signalosome complex amplifies Treg conversion selectively in the TME, which you can read about here and here. Based on these findings, a small molecule inhibitor of Malt1 has now recently entered clinical testing as a cancer treatment.
We are now seeking to identify the local environmental factors, the signaling events, and the epigenetic changes that underlie this localized pro-inflammatory conversion of Treg cells.
Memory and effector cell fate heterogeneity in the naïve T cell compartment
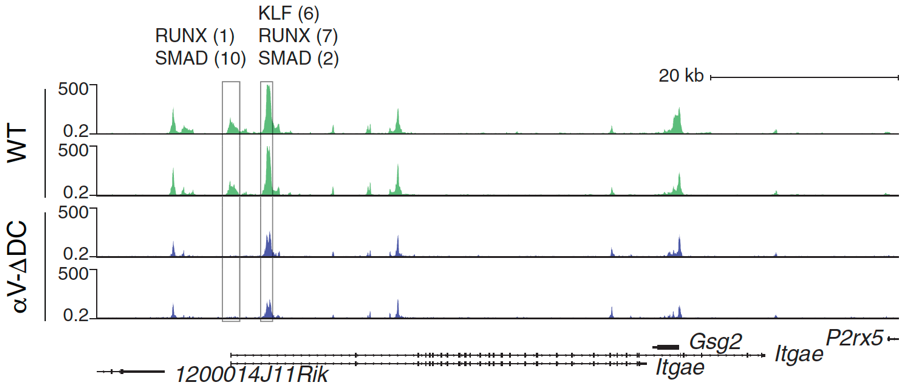 A widespread assumption is that naïve T cells are homogeneous in terms of their capacity to form short-lived effector cells and various kinds of memory cells upon activation through interactions with dendritic cells presenting foreign antigen-derived peptides. Evidence is however mounting that even before foreign antigen encounter, some naïve cells are actively pre-conditioned to adopt specific cell fates. We observed that TGF- signals CD8+ T cells receive in lymph nodes while interacting migratory dendritic cells pre-condition them to form epithelial tissue-resident memory cells in skin upon activation, which you can read about here. Currently, we are seeking to 1) examine the wider spectrum of signals naïve T cells are exposed to ‘at rest’ that bias their later fate, 2) define the context in which they receive these different signals, and 3) identify the intrinsic properties that render naïve T cells preferentially susceptible to some signals more than others. A better understanding of these mechanisms may inform the development of vaccination strategies tailored to produce optimized outcomes in settings where specific T cell effector or memory states are desirable (e.g., during prophylactic versus therapeutic vaccination).
A widespread assumption is that naïve T cells are homogeneous in terms of their capacity to form short-lived effector cells and various kinds of memory cells upon activation through interactions with dendritic cells presenting foreign antigen-derived peptides. Evidence is however mounting that even before foreign antigen encounter, some naïve cells are actively pre-conditioned to adopt specific cell fates. We observed that TGF- signals CD8+ T cells receive in lymph nodes while interacting migratory dendritic cells pre-condition them to form epithelial tissue-resident memory cells in skin upon activation, which you can read about here. Currently, we are seeking to 1) examine the wider spectrum of signals naïve T cells are exposed to ‘at rest’ that bias their later fate, 2) define the context in which they receive these different signals, and 3) identify the intrinsic properties that render naïve T cells preferentially susceptible to some signals more than others. A better understanding of these mechanisms may inform the development of vaccination strategies tailored to produce optimized outcomes in settings where specific T cell effector or memory states are desirable (e.g., during prophylactic versus therapeutic vaccination).
Harnessing the effector function of antiviral bystander T cells against cancer cells
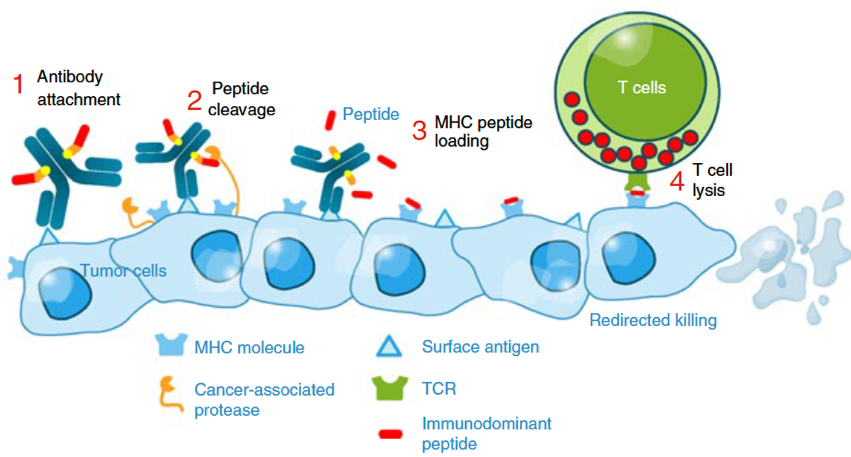 In most solid tumors, the majority of infiltrating CD8+ T cells are bystander cells that do not recognize tumor antigens. Many of these are memory and effector memory cells induced by previous vaccination and past or ongoing viral infections. We are pursuing preclinical development of a novel cancer treatment strategy that seeks to mobilize the cytotoxic function of these antiviral T cells in the TME against cancer cells, using antibody peptide epitope conjugates (APECs), originally developed by Dr. Mark Cobbold and described here. APECs consist of antibodies directed against cell surface proteins overexpressed by cancer cells, to which viral epitopes at attached through a linker that can be cleaved by cancer cell-express proteases. APEC binding to cancer cells is predicted to lead to the release of viral peptides and loading onto empty cell MHC I molecules on the cancer cell surface, causing nearby antiviral bystander T cells to deploy their cytotoxic anti-tumor function. We are seeking to establish the safety of this novel approach to cancer immunotherapy and determine which antiviral T cell subsets are most effectively targeted in human patients.
In most solid tumors, the majority of infiltrating CD8+ T cells are bystander cells that do not recognize tumor antigens. Many of these are memory and effector memory cells induced by previous vaccination and past or ongoing viral infections. We are pursuing preclinical development of a novel cancer treatment strategy that seeks to mobilize the cytotoxic function of these antiviral T cells in the TME against cancer cells, using antibody peptide epitope conjugates (APECs), originally developed by Dr. Mark Cobbold and described here. APECs consist of antibodies directed against cell surface proteins overexpressed by cancer cells, to which viral epitopes at attached through a linker that can be cleaved by cancer cell-express proteases. APEC binding to cancer cells is predicted to lead to the release of viral peptides and loading onto empty cell MHC I molecules on the cancer cell surface, causing nearby antiviral bystander T cells to deploy their cytotoxic anti-tumor function. We are seeking to establish the safety of this novel approach to cancer immunotherapy and determine which antiviral T cell subsets are most effectively targeted in human patients.